This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What are protein domains?
A protein domain is a portion of a protein or polypeptide chain that is characterized by a specific folding pattern or structural motif. These domains can vary greatly in both length and amino acid type, and different domains can be found within a single protein to give that protein a unique function. Protein domains for a specific protein may or may not be conserved amongst different species, and they can offer insight into which regions of a protein are vital to its proper functioning [1,2].
What domains are found in CNBP protein?
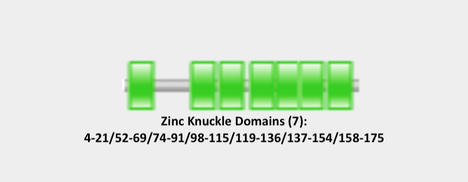
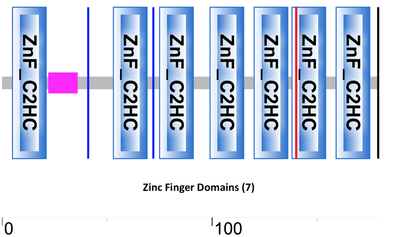
Pfam, SMART InterPro, PROSITE, and GO were used to characterize the protein domains of the CNBP protein. All the databases confirmed seven separate zinc-finger or zinc-knuckle domains in the same residues of CNBP, with the results of Pfam and SMART depicted below (Figures 1 & 2). Six of the seven zinc-finger domains come after the first intron of CNBP (indicated by the blue line in Figure 2), which is the region of the CNBP associated with sequence repeats and the cause of myotonic dystrophy type 2 (DM2).
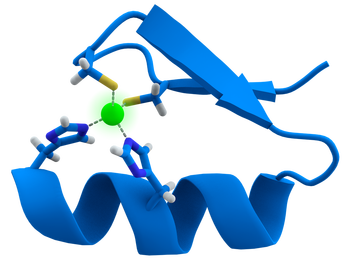
Zinc-finger (ZnF) domains are regions of a protein that contain finger-like, amino acids extensions that coordinate zinc ions or other metal ions to stabilize the fold. Though not all ZnF domains use metal ions to stabilize their folds, ZnF domains are still very stable structures that do not usually undergo conformational changes. They are a commonly found protein domain with a variety of specialized functions including regulation of transcription, translation, and mRNA trafficking. Often times ZnF domains will be found in clusters where the fingers of each domain may have different binding specificities [3]. Depicted below is an image of a ZnF motif comprised of two histidines and two cysteine residues to stabilize a zinc ion:
How conserved are CNBP domains across species?
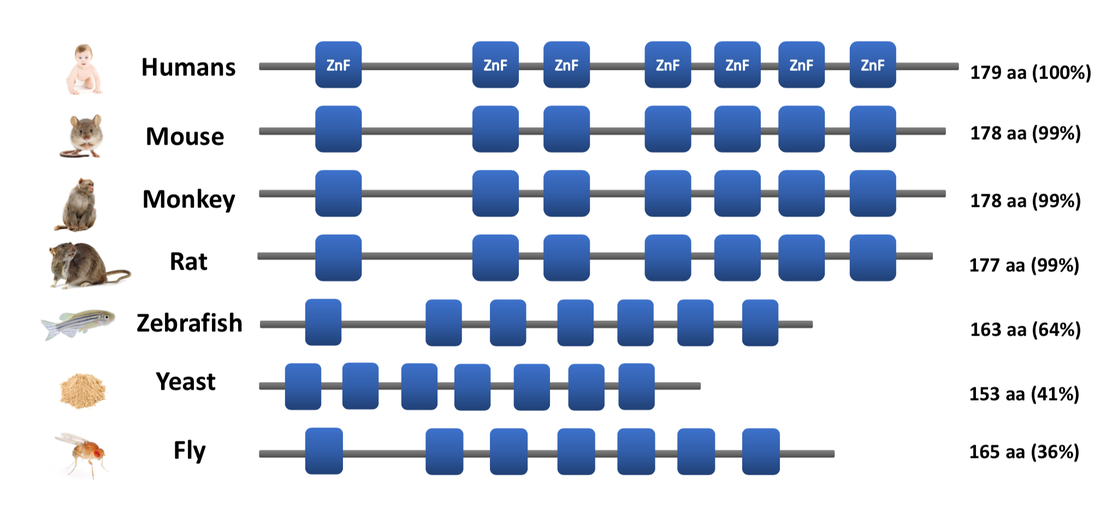
The ZnF domains appear to be a key feature of the CNBP protein. All the species depicted contain seven distinct ZnF domains, though they differ in where they lie within the gene due to differences in length. The ZnF domains are almost perfected conserved across the first four species of mammals, therefore suggesting that each domain may play a vital role within each organism. Even though Drosophila melanogaster only has 36% percent identity to humans, it too contains seven ZnF domains.
Discussion
Zinc-finger domains are small protein domains with fingerlike amino acid protrusions that coordinate zinc ions. They form stable amino acid structures, perform a variety of functions including DNA and RNA binding, and generally are found in clusters, like in CNBP. CNBP contains seven ZnF domains, with six of the seven domains coming after the tetranucleotide repeat sequences that are found in the first intron of DM2 patients. ZnF domains are highly conserved across species that carry the CNBP gene, thus suggesting that these domains plays a critical role in CNBP function. The fact the CNBP protein is so highly conserved amongst muscular species also suggests that this protein may have functions unique to the regulation of skeletal muscle function and play a role in DM2.
References
- Protein domain. (n.d.). Retrieved March 16, 2018, from https://www.biology-online.org/dictionary/Protein_domain
- What are protein domains? (2016, July 20). Retrieved March 16, 2018, from https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
- Laity, J. H., Lee, B. M., & Wright, P. E. (2001). Zinc finger proteins: New insights into structural and functional diversity. Current Opinion in Structural Biology, 11(1), 39-46. doi:10.1016/s0959-440x(00)00167-6
Header: https://i1.wp.com/www.artofthecell.com/wp-content/uploads/2014/10/John-Liebler-RNA-Polymerase.jpg?ssl=1